 The elements in each period were linked by tie lines with one or more elements in the following period. The table contained a period of 2 elements, two of 8 elements, two of 18 elements, one of 32 elements, and an incomplete period. The table constituted periods of increasing length between the noble gases. This was more determined by the electronic structure of atoms proposed by Niels Bohr in 1922. The “short-period” form of the periodic table, with Groups 0, I, II,…VIII, attained prominence and popularity, staying in relevance almost until 1930. Post discovery of the noble gases helium, neon, argon, krypton, radon, and xenon by Lord Rayleigh (John William Strutt) and Sir William Ramsay in 1894, Mendeleyev along with others recommended the addition of a new “zero” group to the atomic table of elements, that can accommodate them. 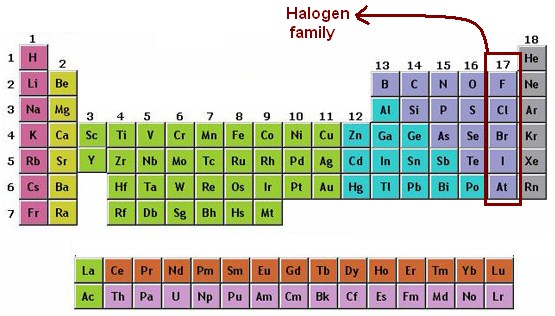
This was because they exhibited certain common features to those on hafnium (72), tantalum (73), and tungsten (74). They used to be categorized under groups IVa, Va, and VIa, respectively. This was the era when thorium (90), protactinium (91), and uranium (92) did not get clarity on whether they were part of the actinide series. Short-period form of Periodic system of elements, 1930 The first and second periods of seven were later differentiated through using of the letters “a” and “b” attached to the group symbols, which were the Roman numerals. He came up with a table of eight columns that was derived by fragmenting every long period into a period of seven, an eighth group containing the three central elements (such as iron, cobalt, nickel Mendeleyev also included copper, and did not categorize it in Group I), and a second period of seven. The major enhancement was the exact positioning of 17 elements, in a way altered positioning. This is in turn followed by three incomplete periods.īack in 1871 Mendeleyev presented a paper that stood as an improved and altered pattern, with a 17-group table. This ranged from potassium to bromine and rubidium to iodine and followed two partial periods of seven elements each (lithium to fluorine and sodium to chlorine). In 1869, Mendeleyev’s periodic table covered 17 columns, with two almost complete periods (sequences) of elements. And over the years, this table has been labeled as a Periodic Table or the atomic table of elements. However, at first, it was hard to organize elements into different groups thus, it took rearranging multiple times. In the year 1869, a Russian Scientist started categorizing available elements into groups and categories based on their atomic numbers and chemical formulas, as well as common similarities between elements, which are called Periodic Trends. Before we get into the topic, let us first understand the History of the Periodic Table. Let us first begin with the understanding of the periodic table. 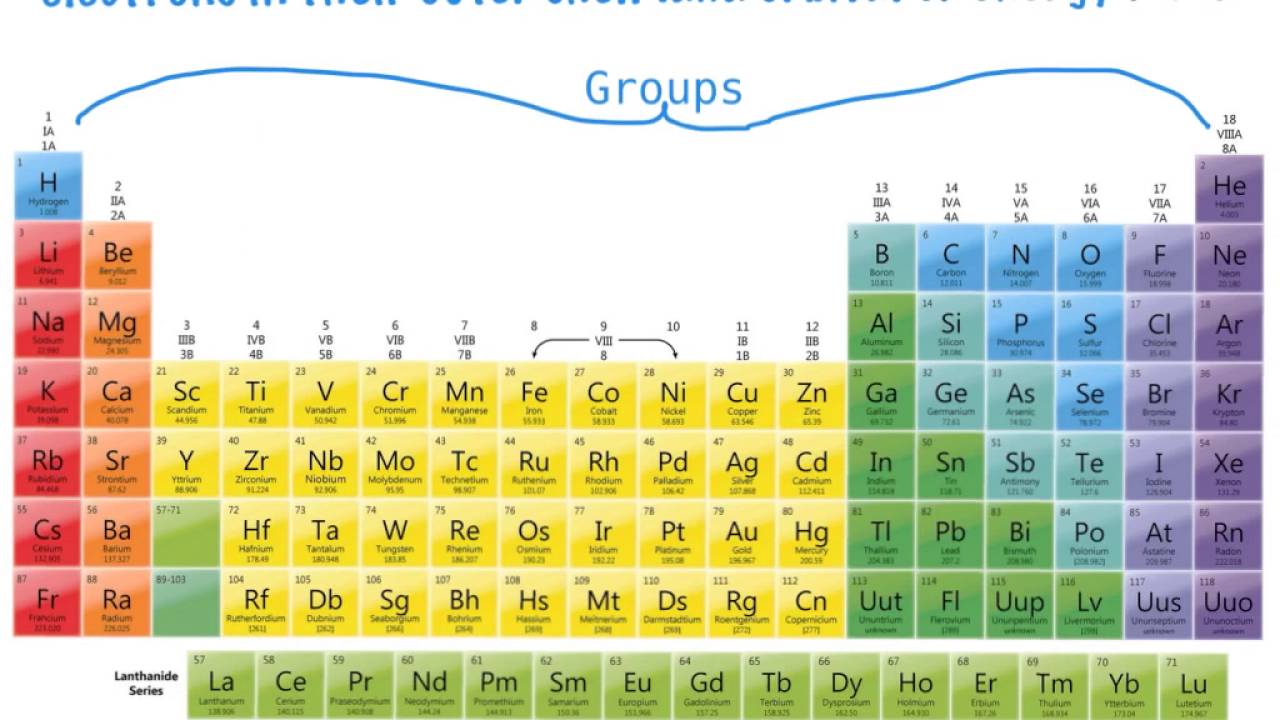
In this article, let us address the above contents, understanding the atomic table of elements in a broader sense What is a Periodic Table?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
